EasyTear (Gen2A) Impervious Isolation Gown
Recommended for use when expected risk of exposure to fluid is HIGH, such as: Suturing, blood draw, inserting I.V. lines, specimen handling, drawing arterial blood. ICU, Med/Surg Unit, Hyperbaric & Dialysis Units, Paramedics and First Responders.
Impervious to liquids and bodily fluids.
Material: LDPE (FDA 177.1520/(EU) No.10/2011 compliant)
Color: Transparent tinted blue
Dimension: 780x1260mm
Thickness: 0.033mm
Packing: 50pcs folded per stack in polybag
Carton Size: 39x33x38cm
GW (carton): 8.55kg
Qty/carton: 100 pcs
MOQ: 20,000 pcs
Certification: ISO 13485:2016, CE, AAMI PB70 Level 3, RoHS
Features:
Patented ‘EasyTear’ waist tie design
Patented ‘EasyTear’ enclosed back design
Hood with drawtape
Patented EasyTear gown with hood for maximum protection against particulates
What is the difference between fluid resistant and impervious isolation gowns?
Isolation gowns are all designed to do the same thing, prevent infectious diseases and pathogens from contaminating your clothes and spreading to others.
There are two types of isolation gowns; fluid resistant isolation gowns and impervious isolation gowns.
Fluid resistant gowns will repel light fluids such as blood and urine, but only for a short period, as these can soak into the gown after time.
Impervious isolation gowns can withstand larger amounts of fluid and will not seep through the material and contaminate the clothing and wearer.
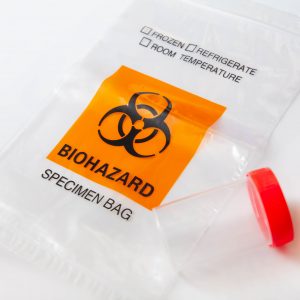
The ANSI/AAMI PB70 standard includes four standard tests to evaluate the barrier effectiveness of surgical gowns, isolation gowns, and surgical drapes. Based on the results of these standardized tests, four levels of barrier performance are defined, with Level 1 being the lowest level of protection, and Level 4 being the highest level of protection. Table 3 summarizes the requirements of ANSI/AAMI PB70:2012 regarding the classification of barrier performance of surgical gowns, isolation gowns, and surgical drapes.
ANSI/AAMI PB 70:12 classification of barrier performance of surgical gowns, other protective apparel, surgical drapes and drape accessories
As indicated in the above table, the requirements for levels 1, 2, and 3 have specific test requirements associated with them. Only Level 4 gowns are tested for viral penetration resistance, and therefore only Level 4 garments are considered impermeable to viral penetration using ASTM F1671. The gowns complying with the lower levels (Level 1, 2, and 3) cannot be considered impermeable. However, Level 1–3 gowns can provide increasing resistance to liquids.
ANSI/AAMI PB70 Level 1, 2, and 3 surgical/isolation gown-testing requirements only use water as a challenge. Because the surface tension of water is much higher than that of blood, blood can penetrate through fabrics more readily than water. Therefore, no correlation can be made between AATCC water resistance tests (AATCC 42 and AATCC 127) and ASTM F1671 viral penetration tests, and consequently, protection provided by Levels 1, 2, 3 gowns and that of Level 4 gowns. A common misunderstanding among many end-users is that they are protected from blood, body fluids, and other potentially infectious materials (OPIM) when they wear any type of fluid-resistant garment or surgical or isolation gown.